In many countries worldwide including the Philippines, India and South America, lightening your entire face or large areas of your body is a common goal to make one’s self more competitive in the marriage or job market. A lighter complexion is often seen as a way to climb up the social ladder, make new contacts, find a spouse, get a job etc.

In late 2009 Dominican-born Chicago Cubs baseball player Sammy Sosa showed up at the Latin Grammies with skin lightened by several shades. Sosa admitted to using creams to lighten his skin as well as getting bleaching treatments.
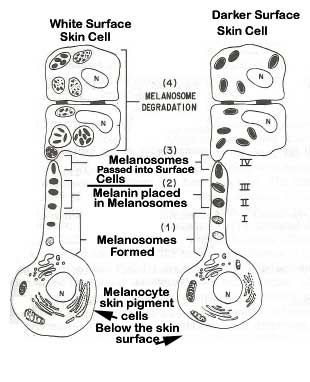
Pigment cells in the skin called melanocytes produce skin pigmentation called melanin from the amino acid tyrosine. The reaction is stimulated by genetics, sun exposure, skin inflammation or irritation and the aging process. The melanocytes put the pigment in packets called melanosomes and transfer them into skin surface cells.
Darker skinned individuals have larger sized and a greater number of melanosomes in their skin surface cells.
Hydroquinone, a molecule discovered in the late 1800s and initially used to develop photographs, can block this reaction. The ability of hydroquinone to depigment skin was first noted in 1940 in wearers of rubber gloves made with compounds related to hydroquinone. Skin discoloration (the opposite of depigmentation) from chronic over application of hydroquinone was first noted in 1960. We call this onchronosis.
Later it was discovered that the addition of vitamin A derivatives and topical steroids allowed patients to tolerate higher concentrations of hydroquinone and lighten skin more quickly. The combination was so effective that it could create complete white patches of skin on an Afro-American no matter how dark their baseline skin color was. The problem was application of 3 different creams a day was cumbersome. At that time if you combined them they would interact with room air and the resulting product (triple bleaching cream) had a very short shelf life. By the late 1990s that problem was solved so stable triple skin bleaching creams became available. I would use them to prepare patients for laser skin resurfacing and then restart the creams a week or 2 after the laser treatment. This regimen prevented the temporary increased pigmentation we used to see after laser skin resurfacing treatments.
In 1982, the FDA published a rule to propose that over the counter skin bleaching drug products containing 1.5 to 2 percent hydroquinone be generally recognized as safe and effective (GRASE). The higher concentrations of 4% or more required a prescription. In 2006 research revealed that hydroquinone taken orally by mice or rats could cause cancer. Note that humans only apply it to the skin surface at much lower doses. They do not eat it. It also has been linked with a medical condition in humans known as ochronosis (skin darkening and disfiguration) when applied topically. At that time the FDA requested more scientific testing on hydroquinone because doctors and patients protested against an outright ban. Those tests began in 2009 despite a hydroquinone ban in Britain beginning in 2001. In 2011 sales of 4% hydroquinone products was banned in the state of Texas.
Less than 10 years ago a commercially available triple bleaching cream called Triluma became available by prescription. On 11/17/10 the FDA recalled specific batches of Triluma, “Firm was notified by supplier of a subpotent active ingredient”. That is not the same as a complete ban and the product can still be used. It contains 4% hydroquinone and therefore requires a prescription.
Directions for usage of both over the counter and prescription strengths clearly state that hydroquinone is to be used for only short term (4-6 months) and should be stopped if no result is seen by that time.
With prolonged exposure to hydroquinone, even the weaker over the counter formulations, there is progressive sooty darkening, inflammation, irritation, thickening and hyperpigmentation of the skin (darkened bluish/gray patches of skin). This condition is called onchronosis and individuals with darker complexions are at higher risk of getting it after prolonged use. It can occur with frequent applications in less than a year. For this reason it is recommended that hydroquinone be used for spot treatment and not all over the skin and for short durations. Allergic reactions to hydroquinone can cause dermatitis, which may lead to dark or reddened, inflamed and scaly patches on the skin.
Episode of Dr. Oz highlighting the problems with the over use of hydroquinone containing products.
For this reason it is recommended that hydroquinone be used for spot treatment and not all over the skin and for short durations. Typically, skin lightening can be seen in about four weeks. Once the desired effect is achieved, the treatment may be used less frequently to maintain results.
Those with sensitive skin, should test the hydroquinone product on a small patch of skin before applying to large areas. If no adverse reactions are seen within 24 hours, the treatment can be continued. Mild side effects such as itching, stinging or slight redness are usually temporary. More serious side effects include: burning, itching, crusting or swelling, and unusual skin discoloration one should stop using the product immediately and see a doctor if any of these reactions occur. The skin develops tachyphylaxis to hydroquinone (over time more hydroquinone is required to get the same effect) so 1-month “holidays” without applying hydroquinone are recommended in order to maintain its effectiveness.
Lumixyl™ is a non-toxic, non-irritating, synthetic that can also lighten skin color or even out skin surface pigmentation by blocking the melanin producing enzymes. Lumixyl is an over the counter product sold through professional channels, whereas Triluma requires a prescription from a doctor.
Studies show that Lumixyl™ can reduce melanin synthesis by as much as 40 percent compared to treatments with hydroquinone, which reduced production by only 7 percent in the same study.
Other skin-lightening options include:
* Arbutin: Derived from the leaves of blueberry, bearberry, cranberry or mulberry shrubs, or from certain genus of pear, arbutin contains the natural form of hydroquinone.
* Azelaic acid: Most commonly, this ingredient treats acne, but recent studies have shown it benefits skin discoloration, too. Azelaic acid is made from wheat, rye or barley, and is usually found in creams at a concentration of 20 percent.
* Kojic acid: Derived from fungus and fermented rice, kojic acid also inhibits melanin production. Kojic acid oxidizes quickly and will turn brown if left exposed to air. Check the expiration date and be sure to store kojic acid products in a cool, dark place, away from heat or light.
* Licorice extract: A tyrosinase inhibitor, licorice extract is a potent skin lightener that disrupts skin’s melanin synthesis and also has anti-inflammatory qualities.
* Mild alpha hydroxy acids like Malic acid act as exfoliants. By sloughing off dead skin cells, they take away extra pigment and pigment cells as well.
* Dermamelan is a mixture of mild chemical peels, sunscreen, vitamin C and a natural hydroquinone like substance (Kojic Acid, Phytic Acid, Ascorbic Acid, Titanium Dioxide, Arbutine and Retinyl Palmitate) that is first applied as a mask in the doctor’s office. The patient goes home with the mask in place and then applies a cream of the material on a daily basis for several months. Skin lightening is visible within 2 to 8 weeks.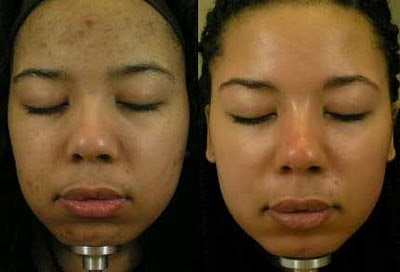
Before and After Dermamelan Treatment
* Cosmelan1 is a similar cream containing Azelaic Acid, Kojic Acid, Phytic Acid, Ascorbic Acid, Arbutin, and Titanium Dioxide. It is applied in the doctor’s office as a mask that is left on for a total of 4-8 hours depending on your skin type. The mask is then washed off with water and a neutral cleanser. Cosmelan2 which lacks the Arbutin is then applied at home twice a day for 2 weeks then once a day for up to a year as a maintenance cream. Redness with skin flaking and itching may be seen in the first few days of this treatment regimen. The effects begin within the first week and peak 4 to 6 weeks into the regimen.
* Benoquin (Monobenzone) 20% is a topical cream that is recommended for lightening the skin edges around patches of vitilgo to decrease the contrast between pigmented and depigmented areas of skin. For you chemists out there Monobenzone is the monobenzyl ether of hydroquinone. It causes permanent discoloration i.e it kills pigment cells and so is not recommended for sun spots, age spots, moles, freckles or skin discoloration caused by hormones, medicine, perfumes, pregnancy, or skin trauma. It should not be considered as a substitute for hydroquinone or as a cosmetic skin bleach. It may take up to 4 months before the full benefit of this drug takes effect. Once the desired skin color is achieved, this medication is applied only as needed to maintain your new skin color (usually 2 times a week). Problems that have been encountered with this medication include leaking of the action to adjacent untreated areas causing permanent skin discoloration of areas that you did not want to treat and severe allergic reactions.
All of the treatments described above either prevent melanin formation (which takes a week or 2 to become effective and a month or more to see results) or removes the outer surface of the skin. Lignin peroxidase is an enzyme that breaks down existing melanin without affecting melanin biosynthesis or blocking tyrosinase so its results can be seen right away. It is produced by submerged fermentation of the fungus Phanerochaete chrysosporium 3 and then purified from the fermented liquid medium. The safety of lignin peroxidase as a skin-lightening active ingredient
has been demonstrated in preclinical studies with doses that are 17,000 times the recommended
dose without prompting any side effects. It does not mutate cells and
is nonirritating to eyes. The potential for skin irritation is very low,
and in studies of 50 subjects each, there were no reports of skin
irritation during acute sensitivity or cumulative sensitivity, or when
used in sensitized skin.
A product containing this is scheduled for release in the United States sometime in 2011.
Some have used glutathione administered intravenously (Glutathione IV) as a skin-whitening agent. It has to be administered intravenously because it is destroyed by acid in the stomach and liver enzymes in the intestines. It may induce a skin-whitening effect by inactivating the enzyme tyrosinase, which is needed in melanin skin pigment production and converts the pigment to the lighter pheomelanin. At very high doses it is unsafe and may result in serious consequences such as the deadly Stevens-Johnson syndrome and toxic epidermal necrolysis; thyroid function derangement; suspected kidney dysfunction, potentially resulting in kidney failure; and severe abdominal pain.
Anything that lightens the skin automatically makes it more sensitive to sun damage including sun induced skin cancers. Also sunlight exposure directly counteracts the effects of skin bleaching creams. So these products should always be used in conjunction with sunscreens. You can get more information about sunscreens at my blog Suntanning and sunscreens
The FDA has banned the sale of mercury-containing creams in the US. Despite that high levels of mercury have been found in foreign-made skin-lightening creams across the country, including Illinois, New York, Minnesota, Virginia and California. The CDC identified a Mexican made skin cream responsible for Mercury exposure in 5 households in California and Virginia. Elevated levels of urine Mercury were found in 9 users of the cream and 6 family members who did not use the cream. The nonusers were exposed through contact with cream users or with contaminated household items. The age of these individuals ranged from 8 months to 67 years. This is particularly alarming because young children are especially susceptible to Mercury poisoning. 6 of the 15 exhibited Mercury poisoning symptoms.
September 26, 2012 Addendum:
Melanozyme is a ligning peroxidase available only in a skin-lightening product known on the market as Elure. The Elure products are presented in a two-sided dispenser with one side containing the Melanozyme component and the other side an activator. Melanozyme alone has little ability to lighten skin, and first needs to be oxidized by hydrogen peroxide (0.012% in the activator) to enter an “activated state.” The activator, which contains a small amount of hydrogen peroxide, is applied to the surface of the skin after the Melanozyme.
Elure has been shown to be better tolerated and more effective than over the counter available 2% hydroquinone. However, more studies are needed to compare the product against stronger concentrations of hydroquinone and other existing treatments, as well as to demonstrate its effectiveness in the treatment of other pigmentary conditions in a broader range of patients. The use of Elure in a combination skin care regimen with hydroquinone and glycolic acid has not been studied, but there is no reason to believe that these products would be incompatible. In fact, a glycolic cleanser that temporarily lowers the pH of the skin prior to application could theoretically enhance the efficacy of the product since Elure is inactivated at normal skin pH.
Allure magazine just named Syneron Medical’s elure™ Lotion the “Best Anti-Ager for Discoloration” in its annual Best of Beauty issue.
This is the second consecutive win for elure in Allure magazine’s Best of Beauty issue. In October 2011, elure™ Lotion and the elure™ Night Cream won the coveted award in the Big Breakthroughs category. Winners are selected based on real, noticeable results.
In addition to the Best of Beauty award, Allure magazine also named elure Lotion one of “50 Beauty Products to Buy Before You Die.”
Aaron Stone MD – Plastic Surgeon Los Angeles
Aaron Stone MD – twitter


![]()

![]()






Good blog! I really love how it is nice on my eyes it is. I am wondering how I might be notified whenever a new post has been made. I've subscribed to your RSS which may do the trick? Have a nice day!